Hydrogen-bonding controlled rigidity of an isoindoline-derived nitroxide spin label for nucleic acids
Dnyaneshwar B. Gophane and Snorri Th. Sigurdsson*
a Department of Chemistry, Science Institute, University of Iceland, Dunhaga 3, 107 Reykjavik, Iceland
Chem. Commun., 2013, 49, 999—1001
[Link: http://pubs.rsc.org/en/content/articlelanding/2013/cc/c2cc36389e#!divAbstract]
Graphical abstract
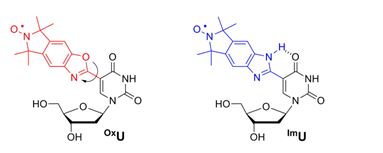
Two new nitroxide-modified nucleosides, OxU and ImU, were synthesized and incorporated into DNA. ImU has lower mobility in duplex DNA due to an intramolecular hydrogen bond.
Abstract
Nucleosides spin-labelled with isoindoline-derived benzimidazole (ImU) and benzoxazole (OxU) moieties were synthesized and incorporated into DNA oligonucleotides. Both labels display limited mobility in duplex DNA but ImU was less mobile, which was attributed to an intramolecular hydrogen bond between the N-H of the imidazole and O4 of the uracil nucleobase.
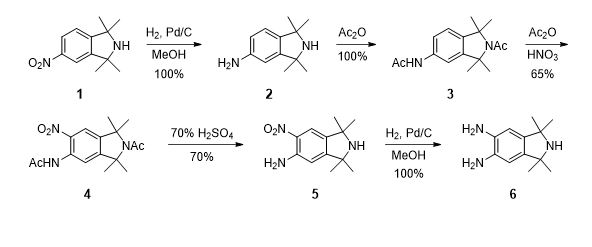
Scheme 1. Literature methods for synthesis of diamino isoindoline 6.
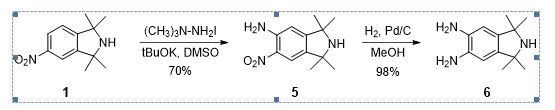
Scheme 2. Improved synthesis of diamino isoindoline 6.
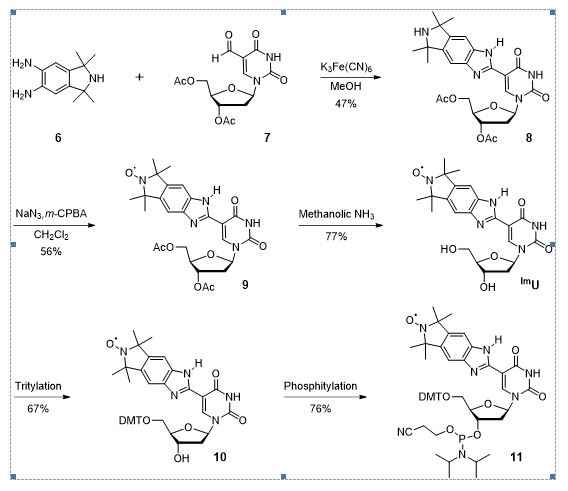
Scheme 3. Synthesis of benzimidazole derivative phosphoramidites 10.
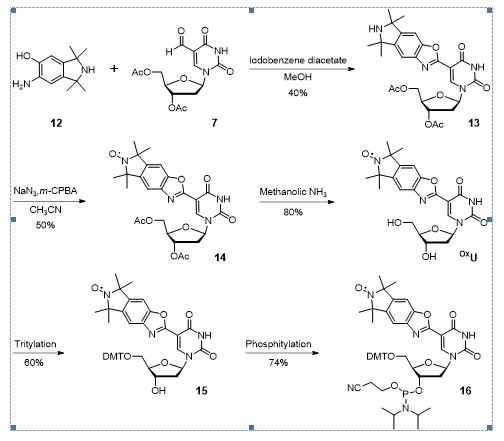
Scheme 4. Synthesis of benzoxazole derivative phosphoramidites 14.
Highligts
- Synthesized novel nitroxide-labelled benzimidazole (ImU) and benzoxazole (OxU) derivatives of 2′-deoxyuridine as spin probes for nucleic acids.
- Both ImU and OxU had limited mobility in duplex DNA, in particular ImU, indicating that rotation around the single bond linking the spin label to the uracil is restricted.
- ImU is the first example of using intramolecular hydrogen-bonding to restrict spin label mobility.
- ImU should not only be a good label for accurate distance measurements in oligonucleotides, but also yield information about the relative orientation of the labels.
ABOUT GUEST BLOGGER
Dr. Dnyaneshwar B. Gophane, Ph. D.
Post doc fellow at Purdue university and university of Iceland
Email, gophane@gmail.com
Dr. Dnyaneshwar Gophane
Dr. Dnyaneshwar B. Gophane completed his B.Sc. (Chemistry) at Anand college of science, Pathardi (Ahmednagar, Maharashtra, India) in 2000 and M.Sc. (Organic Chemistry) at Department of Chemistry, University of Pune (India) in 2003. From 2003 to 2008, he worked in research and development departments of pharmaceutical companies like Dr. Reddy’s Laboratories and Nicholas Piramal India Limited, where he involved in synthesizing novel organic compounds for in vitro and in vivo screening and optimizing process for drug molecule syntheses. In 2008, Dnyaneshwar joined Prof. Sigurdsson’s laboratory for his Ph.D. study at the University of Iceland. His Ph.D. thesis mainly describes syntheses of nitroxide spin-labeled and fluorescent nucleosides and their incorporation into DNA and RNA using phosphoramidite chemistry.
These modified nucleosides are useful probes for studying the structure and dynamics of nucleic acids by EPR and fluorescence spectroscopies. In 2014, after finishing his Ph.D., he worked as post doc fellow in same laboratory and mainly worked on spin labelling of RNA. At the university of Purdue in his second post doc, he was totally dedicated to syntheses of small molecules for anti-cancer activity and modification of cyclic dinucleotides for antibacterial activity. During his research experience, he has authored 8 international publications in peer reviewed journals like Chemical Communications, Chemistry- A European Journal, Journal of organic chemistry and Organic and Biomolecular Chemistry.