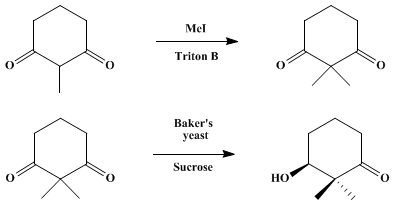

(S)-(+)-3-hydroxy-2,2-dimethylcyclohexanone
bp 85–87°C at 3.7 mm, [α]21D + 23.0° (CHCl3, c 2.0)
The spectral properties of (S)-(+)-3-hydroxy-2,2-dimethylcyclohexanone are as follows:
IR vmax (film) cm−1: 3470 (s), 1705 (s), 1120 (m), 1055 (s), 985 (s), 965 (m);
1H NMR (250 MHz, CDCl3) δ: 1.11 (s, 3 H), 1.15 (s, 3 H), 1.60–1.71 (m, 1 H), 1.76–1.86 (m, 1 H), 1.96–2.05 (m, 2 H), 2.16 (br s, 1 H), 2.35–2.45 (m, 2 H), 3.69 (dd, 1 H, J = 7.6, 2.9);
13C NMR (76 MHz, CDCl3) δ: 19.7, 20.7, 22.9, 29.0, 37.3, 51.3, 77.8, 215.3.
The optical purity of (S)-(+)-3-hydroxy-2,2-dimethylcyclohexanone can be determined by HPLC analysis.
The (
S)-α-methoxy-α-trifluoromethylphenylacetate (MTPA ester) is prepared according to the reported procedure:
3 HPLC analysis (Column, Nucleosil
® 50-5, 25 cm × 4.6 mm; eluant, hexane : THF = 30 : 1, 1.03 mL/min; detected at UV 256 nm) retention time 35.6 min (98.0–99.4%) and 29.6 min (0.6–(2.0%). Therefore, the optical purity is determined to be 96.0–98.8% ee.
Analysis of the MTPA ester of this product by 250 MHz
1H NMR and capillary GLC (12.5 m, 5% methyl silicone column) failed to detect any more of the minor diastereomer than would have been expected from the purity (98% ee) of the MTPA-Cl employed.
NOTE….Intermediate is

2,2-dimethylcyclohexane-1,3-dione bp 92–97°C (4 mm)
37–38°C.
The spectra are as follows: 1H NMR (250 MHz, CDCl3) δ: 1.29 (s, 6 H), 1.93 (5 lines, 2 H, J = 6.5), 2.67 (t, 4 H, J = 6.9); 13C NMR (76 MHz, CDCl3) δ: 18.1, 22.3, 37.4, 61.8, 210.6.
Natural products synthesized from (S)-3-hydroxy-2,2-dimethylcyclohexanone
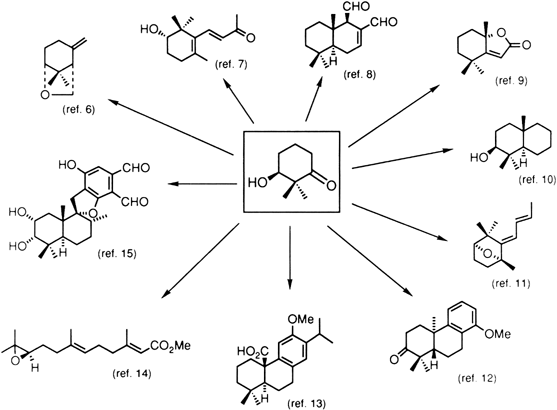
References and Notes
- Department of Agricultural Chemistry, The University of Tokyo, Yayoi 1-1-1, Bunkyo-Ku, Tokyo 113, Japan.
- Mekler, A. B.; Ramachandran, S.; Swaminathan, S.; Newman, M. S. Org. Synth., Coll. Vol. V 1973, 743, 3.
- Dale, J. A.; Mosher, H. S. J. Am. Chem. Soc. 1973, 95, 512.
- Kieslich, K. “Microbial Transformations of Non-Steroid Cyclic Compounds;” Georg Thieme; Stuttgart, 1976, pp. 28–31.
- Lu, Y.; Barth, G.; Kieslich, K.; Strong, P. D.; Duax, W. L.; Djerassi, C. J. Org. Chem. 1983, 48, 4549.
- Mori, K.; Mori, H. Tetrahedron 1985, 41, 5487.
- Yanai, M.; Sugai, T.; Mori, K. Agric. Biol. Chem. 1985, 49, 2373.
- Mori, K.; Watanabe, H. Tetrahedron 1986, 42, 273.
- Mori, K.; Nakazono, Y. Tetrahedron 1986, 42, 283.
- Mori, K.; Mori, H.; Yanai, M. Tetrahedron 1986, 42, 291.
- Mori, K.; Tamura, H. Tetrahedron 1986, 42, 2643.
- Sugai, T.; Tojo, H.; Mori, K. Agric. Biol. Chem. 1986, 50, 3127.
- Mori, K.; Mori, H. Tetrahedron 1986, 42, 5531.
- Mori, K.; Mori, H. Tetrahedron 1987, 43, 4097.
- Mori, K.; Komatsu, M. Liebigs Ann. Chem. 1988, 107.
Read all about Organic Spectroscopy on ORGANIC SPECTROSCOPY INTERNATIONAL 
 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO …..FOR BLOG HOME CLICK HERE
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO …..FOR BLOG HOME CLICK HERE
Join me on Linkedin

Join me on Facebook  FACEBOOK
FACEBOOK
Join me on twitter
 amcrasto@gmail.com
amcrasto@gmail.com