DRUG DELIVERY SYSTEMS
Drug delivery is the method or process of administering a pharmaceutical compound to achieve a therapeutic effect in humans or animals. For the treatment of human diseases, nasal and pulmonary routes of drug delivery are gaining increasing importance. These routes provide promising alternatives to parenteral drug delivery particularly for peptide and protein therapeutics. For this purpose, several drug delivery systems have been formulated and are being investigated for nasal and pulmonary delivery. These include liposomes, proliposomes, microspheres, gels, prodrugs, cyclodextrins, among others. Nanoparticles composed of biodegradable polymers show assurance in fulfilling the stringent requirements placed on these delivery systems, such as ability to be transferred into an aerosol, stability against forces generated during aerosolization, biocompatibility, targeting of specific sites or cell populations in the lung, release of the drug in a predetermined manner, and degradation within an acceptable period of time.
http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3465154/
Drug delivery systems: An updated review
Next……..
Nasal route and drug delivery systems.
Abstract
Nasal drug administration has been used as an alternative route for the systemic availability of drugs restricted to intravenous administration. This is due to the large surface area, porous endothelial membrane, high total blood flow, the avoidance of first-pass metabolism, and ready accessibility. The nasal administration of drugs, including numerous compound, peptide and protein drugs, for systemic medication has been widely investigated in recent years. Drugs are cleared rapidly from the nasal cavity after intranasal administration, resulting in rapid systemic drug absorption. Several approaches are here discussed for increasing the residence time of drug formulations in the nasal cavity, resulting in improved nasal drug absorption. The article highlights the importance and advantages of the drug delivery systems applied via the nasal route, which have bioadhesive properties. Bioadhesive, or more appropriately, mucoadhesive systems have been prepared for both oral and peroral administration in the past. The nasal mucosa presents an ideal site for bioadhesive drug delivery systems. In this review we discuss the effects of microspheres and other bioadhesive drug delivery systems on nasal drug absorption. Drug delivery systems, such as microspheres, liposomes and gels have been demonstrated to have good bioadhesive characteristics and that swell easily when in contact with the nasal mucosa. These drug delivery systems have the ability to control the rate of drug clearance from the nasal cavity as well as protect the drug from enzymatic degradation in nasal secretions. The mechanisms and effectiveness of these drug delivery systems are described in order to guide the development of specific and effective therapies for the future development of peptide preparations and other drugs that otherwise should be administered parenterally. As a consequence, bioavailability and residence time of the drugs that are administered via the nasal route can be increased by bioadhesive drug delivery systems. Although the majority of this work involving the use of microspheres, liposomes and gels is limited to the delivery of macromolecules (e.g., insulin and growth hormone), the general principles involved could be applied to other drug candidates. It must be emphasized that many drugs can be absorbed well if the contact time between formulation and the nasal mucosa is optimized.
http://www.ncbi.nlm.nih.gov/pubmed/15230360
Next……..


Biodegradable microspheres for parenteral delivery.
Abstract
Nowadays, emphasis is being laid to development of controlled release dosage forms. Interest in this technology has increased steadily over the past few years. Although oral administration of drugs is a widely accepted route of drug delivery, bioavailability of drug often varies as a result of gastrointestinal absorption, degradation by first-pass effect, and hostile environment of gastrointestinal tract. Transdermal administration for percutaneous absorption of drug is limited by the impermeable nature of the stratum corneum. Ocular and nasal delivery is also unfavorable because of degradation by enzymes present in eye tissues and nasal mucosa. Hence, the parenteral route is the most viable approach in such cases. Of the various ways of achieving long-term parenteral drug delivery, biodegradable microspheres are one of the better means of controlling the release of drug over a long time. Because of the lipidic nature of liposomes, problems such as limited physical stability and difficulty of freeze-drying are encountered. Similarly, for emulsions, stability on long-term basis and in suspensions, rheological changes during filling, injecting, and storage poses limitation. Also, in all these systems, the release rate cannot be tailored to the needs of the patient. Parenteral controlled-release formulations based on biodegradable microspheres can overcome these problems and can control the release of drug over a predetermined time span, usually in the order of days to weeks to months. Various FDA-approved controlled-release parenteral formulations based on these biodegradable microspheres are available on the market, including Lupron Depot Nutropin Depot and Zoladex. This review covers various molecules encapsulated in biodegradable microspheres for parenteral delivery.
http://www.ncbi.nlm.nih.gov/pubmed/16566705


Next……..

Nanopharmaceuticals: A New Perspective of Drug Delivery System
Abstract


Next……..



Drug delivery refers to approaches, formulations, technologies, and systems for transporting a pharmaceutical compound in the body as needed to safely achieve its desired therapeutic effect.[1] It may involve scientific site-targeting within the body, or it might involve facilitating systemic pharmacokinetics; in any case, it is typically concerned with both quantity and duration of drug presence. Drug delivery is often approached via a drug’s chemical formulation, but it may also involve medical devices or drug-device combination products. Drug delivery is a concept heavily integrated with dosage form and route of administration, the latter sometimes even being considered part of the definition.[2]
Drug delivery technologies modify drug release profile, absorption, distribution and elimination for the benefit of improving product efficacy and safety, as well as patient convenience and compliance. Drug release is from: diffusion, degradation, swelling, and affinity-based mechanisms.[3] Most common routes of administration include the preferred non-invasive peroral (through the mouth), topical (skin), transmucosal (nasal, buccal/sublingual, vaginal, ocular and rectal) and inhalation routes.[4][5] Many medications such as peptide and protein, antibody, vaccine and gene based drugs, in general may not be delivered using these routes because they might be susceptible to enzymatic degradation or can not be absorbed into the systemic circulation efficiently due to molecular size and charge issues to be therapeutically effective. For this reason many protein and peptide drugs have to be delivered by injection or a nanoneedle array. For example, many immunizations are based on the delivery of protein drugs and are often done by injection.
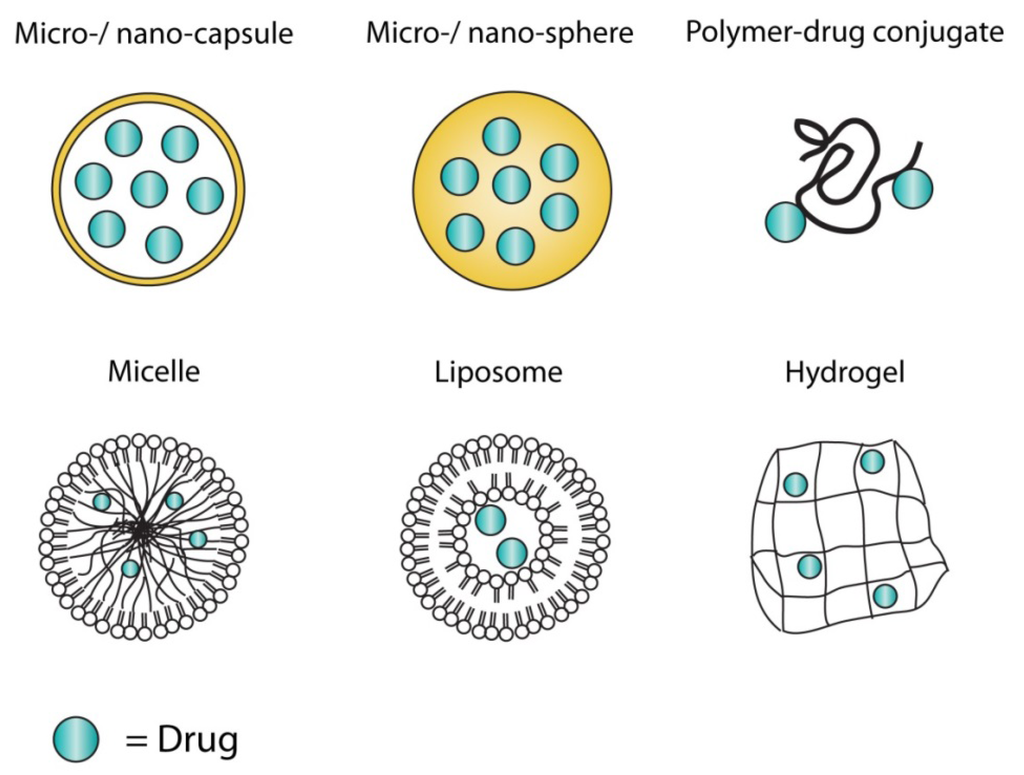
Current efforts in the area of drug delivery include the development of targeted delivery in which the drug is only active in the target area of the body (for example, in cancerous tissues), sustained release formulations in which the drug is released over a period of time in a controlled manner from a formulation, and methods to increase survival of peroral agents which must pass through the stomach’s acidic environment. In order to achieve efficient targeted delivery, the designed system must avoid the host’s defense mechanisms and circulate to its intended site of action.[6] Types of sustained release formulations include liposomes, drug loaded biodegradable microspheres and drug polymer conjugates. Survival of agents as they pass through the stomach typically is an issue for agents which cannot be encased in a solid tablet; one research area has been around the utilization of lipid isolates from the acid-resistant archaea Sulfolobus islandicus, which confers on the order of 10% survival of liposome-encapsulated agents.[7]


See also
- Thin film drug delivery
- Self-microemulsifying drug delivery system
- Acoustic targeted drug delivery
- Neural drug delivery systems
- Drug carrier
- Bovine Submaxillary Mucin Coatings
- Retrometabolic drug design
- Asymmetric membrane capsule
References
- M. N. V. Ravi Kumar (2008), Handbook of Particulate Drug Delivery (2-Volume Set), American Scientific Publishers. ISBN 1-58883-123-X
- “Drug Delivery Systems (definition)”.
- “Drug delivery – definition of drug delivery by Medical dictionary”. TheFreeDictionary.com.
- Wang, NX.; von Recum, HA (2011). “Affinity-Based Drug Delivery”. Macromol Biosci 11: 321–332. doi:10.1002/mabi.201000206.
- “Definition”. Retrieved 2008-05-27.
- “Definition”. Retrieved 2008-05-27.[dead link]
- Bertrand N, Leroux JC. (2011). “The journey of a drug carrier in the body: an anatomo-physiological perspective”. Journal of Controlled Release 161 (2): 152–63.doi:10.1016/j.jconrel.2011.09.098. PMID 22001607.
- Staff (2015). “Acid-friendly Microbe Finds Application in Drug Delivery”. American Laboratory (paper) 47 (9). p. 7. ISSN 0044-7749.
External links



BRAIN

Targeted Drug Delivery to Central Nervous System (CNS) for the Treatment of Neurodegenerative Disorders: Trends and Advances
Central Nervous System Agents in Medicinal Chemistry, 14(1): 43-59.
Author(s): Kritika Goyal, Veena Koul, Yashveer Singh and Akshay Anand.
Affiliation: Neuroscience Research Lab, Department of Neurology, Post Graduate Institute of Medical Education and Research, Chandigarh 160012, India.
Abstract
The treatment of brain diseases has been a major challenge since a long time. Although there are several potent drugs, which are highly therapeutic yet their efficiency is marred due to the presence of the Blood Brain Barrier (BBB). The BBB, which is present at the capillary level regulates and monitors the entry of all small and large molecules entering into the brain. Although this barrier is of immense importance to the brain in terms of safety, it becomes a hindrance when it comes to therapy because the drug molecules are unable to reach the brain. Various biomaterial-based strategies are being developed to overcome the BBB and deliver the drug into the brain. These include polymeric nanoparticles, liposomes, solid-lipid nanoparticles (SLNPs), nanogels, implants, etc. This review provides an overview on CNS disorders, BBB, and various delivery strategies available for biologists engaged in translational neuroscience, to target CNS.

ISSN: 1871-5249 (Print)
Volume 16, 3 Issues, 2016
FREE DOWNLOAD http://www.eurekaselect.com/125799
NEXT……

////////http://www.ncbi.nlm.nih.gov/pubmed/15230360
86,099 total views, 3 views today












