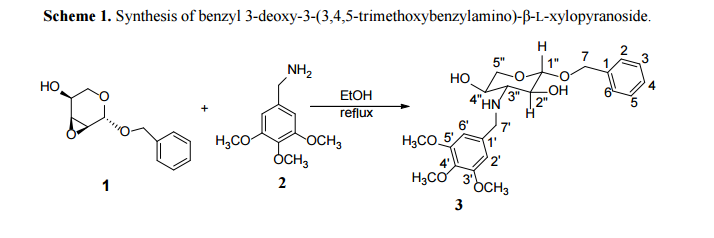
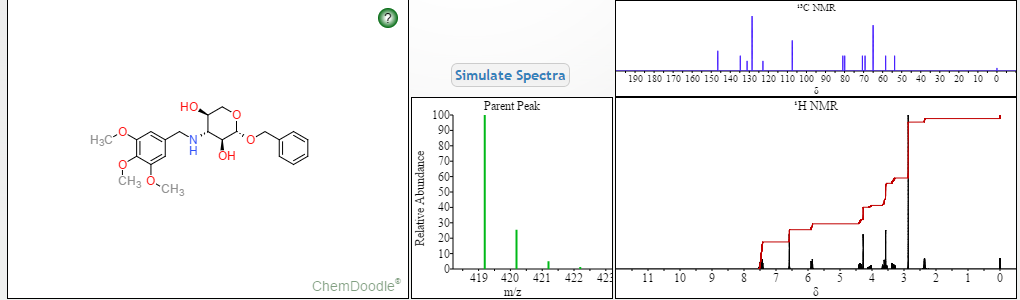
The title compound was synthesized by opening the epoxide of benzyl 2,3-anhydro-β-L-ribopyranoside with 3,4,5-trimethoxybenzylamine (Scheme 1). The three broad peaks in the 1 H-NMR due to one –NH at δ 2.20 ppm, and two –OH at δ 5.00 ppm and 5.26 ppm, disappeared upon D2O exchange. The chemical shifts of the sugar hydrogens, along with COSY and HMBC were used to assign C7, C1″, C2″, C3″, C4″, C5″ and C7′ atoms. The coupling constant between H-1″ and H-2″ on the sugar ring was found to be 7.98 Hz, indicating that the protons at the 1- and 2-positions were in axial positions and that the molecule exists in solution in 1 C4 conformation (Scheme 1). The coupling constant was similar to related analogs [14,15]. The coupling constant between H-2” and H-3” was found to be 9.12 Hz. The coupling constant between the pro-R and pro-S hydrogens on C7 was found to be 12.24 Hz. The 13C had five pairs of atoms with the same chemical shift. There were three pairs of carbon atoms on the 3,4,5-trimethoxybenzyl ring ( two ortho- and two meta-, and two equivalent methoxy groups) that had similar chemical shifts. On the benzyl group, chemical shifts of two pairs of carbon atoms (two ortho- and two meta-) were observed.
PREPN
Benzyl 2,3-anhydro-β-L-ribopyranoside (1) was obtained from L-arabinose in five steps using a previously reported synthetic route [14]. To a mixture of benzyl 2,3-anhydro-β-L-ribopyranoside 1 (0.15 g, 0.68 mmol) and 3,4,5-trimethoxybenzylamine 2 (180 mL, 0.91 mmol) was added ethyl alcohol (3 mL). After refluxing the mixture for 16 h and cooling at room temperature for 12 h, white crystals (needles) formed. Recrystallization from hexane/ethyl acetate mixture (3:2, v/v) produced a pure compound (0.206 g, 72%,
m.p. 158–160 °C);
[α]D 26 +50° (c 1, CHCl3).
C22H29NO7 Calculated: C 62.99; H, 6.97; N, 3.34; O, 26.70 Found: C 62.89; H, 7.01; N, 3.29; O, 26.65
1 H-NMR (400 MHz, Me2SO-d6)
δ 2.20 (bs, 1H, –NH),
2.41 (t, J = 9.12, 7.98 Hz, 1H, H-3),
3.21 (m, 2H),
3.45 (bs, 1H),
3.65 (s, 3H, –OCH3),
3.75 (b, 1H),
3.80 (s, 6H, 2-OCH3),
3.97 (m, 2H),
4.31 (d, J = 7.98 Hz, 1H, H-1),
4.61 (d, J = 12.24 Hz, 1H, –OCH2Ar),
4.80 (d, J = 12.24 Hz, 1H, –OCH2Ar),
5.00 (bs, 1H, –OH), 5.26 (bs, 1H, –OH).
13C-NMR (100 MHz, Me2SO-d6),
δ 53.22 (C-7′),
56.61 (–OCH3),
60.81 (–OCH3),
65.11 (C-3″),
67.37 (C-5″),
70.14 (C-4″),
70.41 (C-7),
73.00 (C-2″),
103.90 (C-1″),
105.80, 128.23, 128.40, 129.00, 136.80, 138.23, 138.93, 153.52.
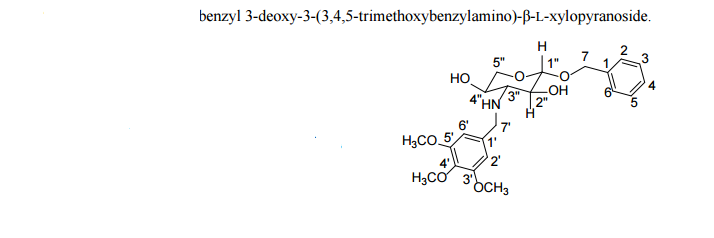
Benzyl 3-deoxy-3-(3,4,5-trimethoxybenzylamino)-β-L-xylopyranoside
//////epoxide ring-opening, 3,4,5-trimethoxybenzylamine, benzyl 2,3-anhydro-β-L-ribopyranoside

















Sorry, the comment form is closed at this time.