
Acting on actin: The mechanism of action of amphidinolide X and amphidinolide J, two relatively small cytotoxic macrolides, has been elucidated. They do not target microtubules and intermediate filaments. The effects observed in A549 and PtK2 cells and the in vitro interaction with actin monomer (G-actin) indicate that these macrolides behave as actin-assembly inhibitors.
Mechanism of Action of the Cytotoxic Macrolides Amphidinolide X and J
DOI: 10.1002/cbic.201100042
Author Information
Email: Prof. Dr. Jaume Vilarrasa (jvilarrasa@ub.edu), Dr. J. Fernando Díaz (i.barasoain@cib.csic.es), Dr. Isabel Barasoain (fer@cib.csic.es)
*Departament de Química Orgànica, Facultat de Química, Universitat de Barcelona, Diagonal 647, 08028 Barcelona (Spain)
Trigili, C., Pera, B., Barbazanges, M., Cossy, J., Meyer, C., Pineda, O., Rodríguez-Escrich, C., Urpí, F., Vilarrasa, J., Díaz, J. F. and Barasoain, I. (2011), Mechanism of Action of the Cytotoxic Macrolides Amphidinolide X and J. ChemBioChem, 12: 1027–1030. doi: 10.1002/cbic.201100042
Corrected by:Corrigendum: Corrigendum: Mechanism of Action of the Cytotoxic Macrolides Amphidinolide X and J
Vol. 12, Issue 9, 1293, Version of Record online: 9 JUN 2011
http://onlinelibrary.wiley.com/doi/10.1002/cbic.201100042/abstract
Others
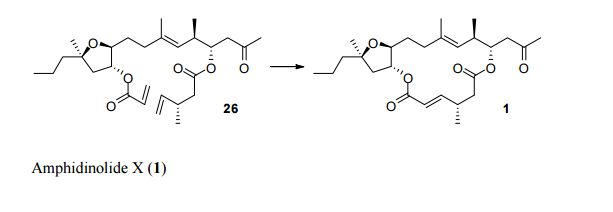
Total Synthesis by Alkene Metathesis: Amphidinolide X (Urpí /Vilarrasa),
To assemble the framework of the cytotoxic macrolide Amphidinolide X (3), Fèlix Urpí and Jaume Vilarrasa of the Universitat de Barcelona devised (Org. Lett. 2008, 10, 5191. DOI: 10.1021/ol8021676) the ring-closing metathesis of the alkenyl silane 1. No Ru catalyst was effective, but the Schrock Mo catalyst worked well.
Total Synthesis of Amphidinolide X & Y
Fürstner
A. Fürstner, E. Kattnig, O. Lepage, J. Am. Chem. Soc. 2006, 128, 9194-9204.
DOI: 10.1021/ja061918e

Another pair of amphidinolides in the bag, Fürstner et al. have completed the synthesis of X (the only member of the series with an even-numbered macrocycle) and Y using a powerful iron catalysed process. Both products (as with most of the family) are cytotoxic, and contain the heavily functionalised THF moiety. This allowed the group to create an intermediate common to both campaigns, starting from a simple epoxide produced from an Sharpless epoxidation.

Treatment of this with n-propyl grignard and catalytic quantities of the iron catalyst generated the allene in a 8:1 dr (this system has been used by the group in other work; see: DOI: 10.1246/cl.2005.624, DOI: 10.1021/ja027190t, DOI: 10.1002/anie.200460504, plus further examples cited in the paper). The allene was then cyclised with silver nitrate and calcium carbonate, returning the DHP, which was augmented to the desired THF via bromoesterification.
This portion of the natural product was coupled using an alkyl Suzuki reaction to the rest of the molecule in both cases, along with macrolactonisation to furnish the major ring system. In amphidinolide Y, a boron-mediated aldol reaction was used to create the 1,4 anti relationship between a pair of hydroxyls in the C1 – C12 fragment, in a 4:1 dr. Inseparable at this point, they carried the mixture through to a diastereoselective methyl grignard addition.

The desired aldol product reacted diastereoselectively with the grignard following the 1,2-anti chelate-cram model, whereas the undesired aldol product reacted with far less control. This section of the synthesis is quite intriguing, and is discussed in far more detail in the paper, which is a truly excellent read.

Figure 1: Scheme 1. (A) Sharpless asymmetric epoxidation of allylic alcohols 1 mediated by Ti(IV)-diethyltartrate (DET) catalyst with alkyl hydroperoxide as terminal oxidant leading to enantioenriched epoxides 2. (B) Preferential attack of the oxygen atom as a function of the stereochemistry of the DET chiral ligand. (C) Schematic representation of the dimeric active catalytic species 3.

Figure 10: Scheme 10. Structure of amphidinolide X 35 and details of the SAE step.
PAPER

http://onlinelibrary.wiley.com/doi/10.1002/anie.200900865/abstract
PAPER

http://onlinelibrary.wiley.com/doi/10.1002/chem.200802069/abstract
////////////Cytotoxic Macrolides, Amphidinolide X and J














Sorry, the comment form is closed at this time.