Valspodar, SDZ-PSC-833, PSC-833, Amdray
P-Glycoprotein (MDR-1; ABCB1) Inhibitors , Multidrug Resistance Modulators
Valspodar is a cyclosporine derivative and a P-glycoprotein inhibitor currently in phase III clinical trials at the National Cancer Institute (NCI) in combination with chemotherapy for the treatment of leukemia. The drug was also being developed in combination with chemotherapy for the treatment of various other types of cancers, however, no recent developments on these trials have been reported.
P-glycoprotein is an ABC-transporter protein that has been implicated in conferring multidrug resistance to tumor cells. In previous trials, valspodar was associated with greater disease-free and overall survival in younger patients (45 years or below), and was shown to significantly increase the cellular uptake of daunorubicin in leukemic blast cells in vivo. However, in a phase III trial examining the drug candidate’s effects on AML in patients at least 60 years of age, valspodar was associated with excessive mortality and complete remission rates were higher in groups not treated with the compound.
Nonimmunosuppressive cyclosporin analog which is a potent multidrug resistance modifier; 7-10 fold more potent than cyclosporin A; a potent P glycoprotein inhibitor; MW 1215.
M.Wt: 1214.62
Formula: C63H111N11O12
CAS : 121584-18-7
IUPAC/Chemical name:
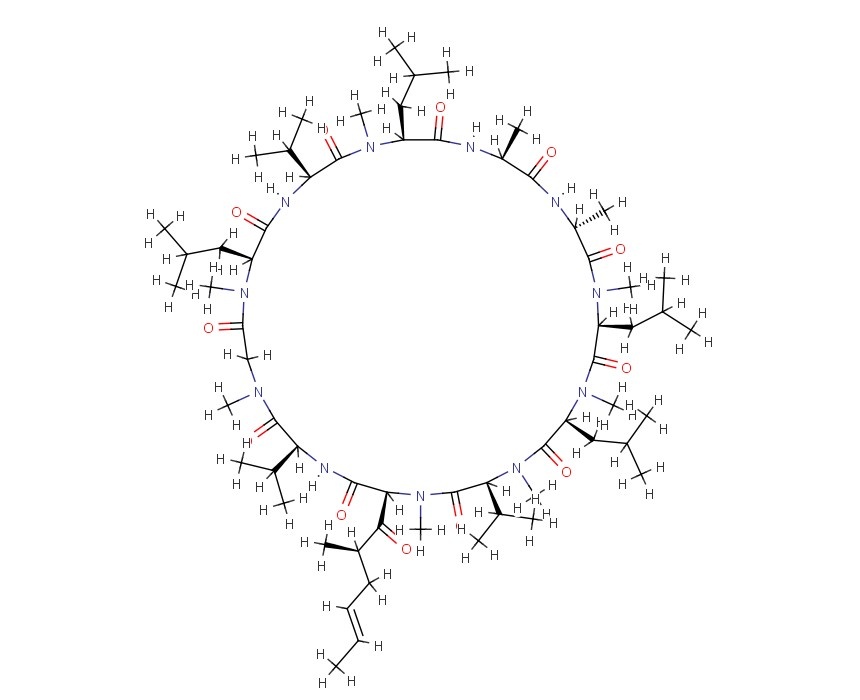
(3S,6S,9S,12R,15S,18S,21S,24S,30S,33S)-6,9,18,24-tetraisobutyl-3,21,30-triisopropyl-1,4,7,10,12,15,19,25,28-nonamethyl-33-((R,E)-2-methylhex-4-enoyl)-1,4,7,10,13,16,19,22,25,28,31-undecaazacyclotritriacontan-2,5,8,11,14,17,20,23,26,29,32-undecaone
6 – [(2S, 4R, 6E)-4-Methyl-2-(methylamino)-3-oxo-6-octenoic acid]-7-L-valine-cyclosporin A; Cyclo [[(2S, 4R, 6E) -4-methyl-2-(methylamino)-3-oxo-6-octenoyl]-L-valyl-N-methylglycyl-N-methyl-L-leucyl-L-valyl-N-methyl-L-leucyl-L- alanyl-D-alanyl-N-methyl-L-leucyl-Nm
[3′-oxo-4-butenyl-4-methyl-Thr1]-[Val2]-cyclosporine
Clinical trials
http://clinicaltrials.gov/search/intervention=psc+833
Synonyms
- 3′-Keto-bmt(1)-val(2)-cyclosporin A
- Amdray
- Psc 833
- PSC-833
- PSC833
- SDZ PSC 833
- Sdz-psc-833
- UNII-Q7ZP55KF3X
- Valspodar
Valspodar or PSC833 is an experimental cancer treatment and chemosensitizer drug.[1] It is a derivative of ciclosporin D.
Its primary use is that of a p-glycoprotein inhibitor. Previous studies in animal models have found it to be effective at preventing cancer cell resistance to chemotherapeutics, but these findings did not translate to clinical success.[2]
Valspodar, also known as PSC-833 is an analogue of cyclosporin-A. Valspodar inhibits p-glycoprotein, the multidrug resistance efflux pump, thereby restoring the retention and activity of some drugs in some drug-resistant tumor cells. This agent also induces caspase-mediated apoptosis.
PSC-833 is a non-immunosuppressive cyclosporin derivative that potently and specifically inhibits P-gp. In vitro experiments indicate that PSC-833interacts directly with P-gp with high affinity and probably interferes with the ATPase activity of P-gp. Studies in multidrug resistant tumor models confirm P-gp as the in vivo target of PSC-833 and demonstrate the ability of PSC-833 to reverse MDR leukemias and solid tumors in mice. Presently,PSC-833 is being evaluated in the clinic.
Valspodar can cause nerve damage.[1]
Valspodar
Synthesis By oxidation of cyclosporin D (I) with N-chlorosuccinimide and dimethylsulfide in toluene (1) Scheme 1 Description alpha (20, D) -..?. 255.1 (c 0.5, CHCl3) Manufacturer Sandoz Pharmaceuticals Corp (US).. . References 1 Bollinger, P., B flounder sterli, JJ, Borel, J.-F., Krieger, M., Payne, TG, Traber, RP, Wenger, R. (Sandoz AG; Sandoz Patent GmbH; Sandoz Erfindungen VmbH ). Cyclosporins and their use as pharmaceuticals.
AU 8817679, EP 296122, JP 89045396. AU 8817679; EP 0296122; JP 1989045396; JP 1996048696; US 5525590
……………………………..
-
The cyclosporins comprise a class of structurally distinctive, cyclic, poly-N-methylated undecapeptides, generally possessing pharmacological, in particular immunosuppressive, anti-inflammatory and/or anti-parasitic activity, each to a greater or lesser degree. The first of the cyclosproins to be isolated was the naturally occurring fungal metabolite Ciclosporin or Cyclosporine, also known as cyclosporin A and now commercially available under the Registered Trade Mark SANDIMMUN®. Ciclosporin is the cyclosporin of formula A
wherein -MeBmt- represents the N-methyl-(4R)-4-but-2E-en-1-yl-4-methyl-(L)threonyl residue of formula B
in which -x-y- is trans -CH=CH- and the positive 2′, 3′ and 4′ have the configuration S, R and R respectively.
-
Since the original discovery of Ciclosporin, a wide variety of naturally occurring cyclosporins have been isolated and identified and many further non-natural cyclosporins have been prepared by total- or semi-synthetic means or by the application of modified culture techniques. The class comprised by the cyclosporins is thus now substantial and includes, for example, the naturally occurring cyclosporins A through Z [c.f. Traber et al. 1, Helv. Chim. Acta, 60, 1247-1255 (1977); Traber et al. 2, Helv. Chim. Acta, 65, 1655-1667 (1982); Kobel et al., Europ. J. Applied Microbiology and Biotechnology 14, 273-240 (1982); and von Wartburg et al. Progress in Allergy, 38, 28-45 (1986)], as well as various non-natural cyclosporin derivatives and artificial or synthetic cyclosporins including the dihydro- and iso-cyclosporins [in which the moiety -x-y- of the -MeBmt- residue (Formula B above) is saturated to give -x-y- = -CH₂-CH₂- / the linkage of the residue -MeBmt- to the residue at the 11-position of the cyclosporin molecule (Formula A above) is via the 3′-O-atom rather than the α-N-atom]; derivatised cyclosporins (e.g. in which the 3′-O-atom of the -MeBmt- residue is acylated or a further substituent is introduced at the α-carbon atom of the sarcosyl residue at the 3-position); cyclosporins in which the -MeBmt- residue is present in isomeric form (e.g. in which the configuration across positions 6′ and 7′ of the -MeBmt- residue is cis rather than trans); and cyclosporins wherein variant amino acids are incorporated at specific positions within the peptide sequence employing e.g. the total synthetic method for the production of cyclosporins developed by R. Wenger – see e.g. Traber et al. 1, Traber et al. 2 and Kobel et al. loc. cit.; U.S. Patents Nos 4 108 985, 4 210 581, 4 220 641, 4 288 431, 4 554 351 and 4 396 542; European Patent Publications Nos. 0 034 567 and 0 056 782; International Patent Publication No. WO 86/02080; Wenger 1, Transpl. Proc. 15, Suppl. 1:2230 (1983); Wenger 2, Angew. Chem. Int. Ed., 24, 77 (1985); and Wenger 3, Progress in the Chemistry of Organic Natural Products 50, 123 (1986).
-
The class comprised by the cyclosporins is thus now very large indeed and includes, for example [Thr]²-, [Val]²-, [Nva]²- and [Nva]²-[Nva]⁵-Ciclosporin (also known as cyclosporins C, D, G and M respectively), [3-O-acetyl-MeBmt]¹-Ciclosporin (also known as cyclosporin A acetate), [Dihydro-MeBmt]¹-[Val]²-Ciclosporin (also known as dihydro-cyclosporin D), [Iso-MeBmt]¹-[Nva]²-Ciclosporin (also known as isocyclosporin G), [(D)Ser]⁸-Ciclosporin, [MeIle]¹¹-Ciclosporin, [(D)MeVal]¹¹-Ciclosporin (also known as cyclosporin H), [MeAla]⁶-Ciclosporin, [(D)Pro]³-Ciclosporin and so on.
-
[In accordance with conventional nomenclature for cyclosporins, these are defined throughout the present specification and claims by reference to the structure of Ciclosporin (i.e. Cyclosporin A). This is done by first indicating the amino acid residues present which differ from those present in Ciclosporin (e.g. “[(D)Pro]³” to indicate that the cyclosporin in question has a -(D)Pro- rather than -Sar- residue at the 3-position) and then applying the term “Ciclosporin” to characterise remaining residues which are identical to those present in Ciclosporin.
-
The residue -MeBmt- at position 1 in Ciclosporin was unknown before the discovery of the cyclosporins. This residue and variants or modifications of it, e.g. as described below, are thus generally characteristic of the cyclosporins. In general, variants or alternatives to [MeBmt]¹ are defined by reference to the -MeBmt- structure. Thus for dihydrocyclosporins in which the moiety -x-y- (see formula B above) is reduced to -CH₂-CH₂-, the residue at the 1-position is defined as “-dihydro-MeBmt-“. Where the configuration across the moiety -x-y- is cis rather than trans, the resulting residue is defined as “-cis-MeBmt-“.
-
Where portions of the -MeBmt- residue are deleted, this is indicated by defining the position of the deletion, employing the qualifier “des” to indicate deletion, and then defining the group or atom omitted, prior to the determinant “-MeBmt-“, “-dihydro-MeBmt-“, “-cis-MeBmt-” etc.. Thus “-N-desmethyl-MeBmt-“, “-3′-desoxy-MeBmt-“, and “-3′-desoxy-4′-desmethyl-MeBmt-” are the residues of Formula B¹, B² and B³ respectively:
B¹ – X = CH₃, Y = OH, Z = H.
B² – X = CH₃, Y = H, Z = CH₃.
B³ – X = H, Y = H, Z = CH₃. -
Where positions or groups, e.g. in -MeBmt-, are substituted this is represented in conventional manner by defining the position and nature of the substitution. Thus -3′-O-acetyl-MeBmt- is the residue of formula B in which the 3′-OH group is acetylated (3′-O-COCH₃). Where substituents of groups, in e.g. -MeBmt-, are replaced, this is done by i) indicating the position of the replaced group by “des-terminology” as described above and ii) defining the replacing group. Thus -7′-desmethyl-7′-phenyl-MeBmt- is the residue of formula B above in which the terminal (8′) methyl group is replaced by phenyl. 3′-Desoxy-3′-oxo-MeBmt- is the residue of formula B above in which the 3′-OH group is replaced by =O.
-
In addition, amino acid residues referred to by abbreviation, e.g. -Ala-, -MeVal-, -αAbu- etc… are, in accordance with conventional practice, to be understood as having the (L)-configuration unless otherwise indicated, e.g. as in the case of “-(D)Ala-“. Residue abbreviations preceded by “Me” as in the case of “-MeLeu-“, represent α-N-methylated residues. Individual residues of the cyclosporin molecule are numbered, as in the art, clockwise and starting with the residue -MeBmt-, -dihydro-MeBmt- etc. … in position 1. The same numerical sequence is employed throughout the present specification and claims.]
-
[0010]Because of their unique pharmaceutical potential, the cyclosporins have attracted very considerable attention, not only in medical and academic circles, but also in the lay press. Cyclosporin itself is now commonly employed in the prevention of rejection following allogenic organ, e.g. heart, heart-lung, kidney and bone-marrow transplant, as well as, more recently, in the treatment of various auto-immune and related diseases and conditions. Extensive work has also been performed to investigate potential utility in the treatment of various parasitic diseases and infections, for example coccidiomycosis, malaria and schistosomiasis. Reports of investigative work into the potential utility of the very many other known cyclosporins in these or related indications now abound in the literature.
………………………………
References
- Wilkes, Gail; Ades, Terri B. (2004). Consumers Guide to Cancer Drugs. Jones & Bartlett Learning. p. 226. ISBN 9780763722548. Retrieved 29 May 2013.
- Tao, Jian’guo; Sotomayor, Eduardo. (2012). Hematologic Cancers: From Molecular Pathobiology to Targeted Therapeutics. Springer. p. 335. ISBN 9789400750289.
- PSC-833Drugs Fut 1995, 20(10): 1010
- US 5525590
- Synthesis of [S-[1-14C]Val(7)]VALSPODAR application of (+)/(-)-[13,14Cn]BABS and (+)/(-)-[13,14Cn]DPMGBS, part 4J Label Compd Radiopharm 2000, 43(3): 205
- WO 2006013094
- WO 2005013947
- WO 2002098418
- WO 1999017757
- Pharmaceutical Research, 2001 , vol. 18, 2 pg. 183 – 190
- US2003/158097 A1
- Valspodar; EP-B1 0 296 122:
- WO 94/07858


















Sorry, the comment form is closed at this time.