
niraparib
MK-4827
MK-4827 Formula: C19H20N4O
MK-4827 Storage: at -20 ℃ 2 years
MK-4827 CAS No.: 1038915-60-4
MK-4827 is an inhibitor of PARP 1 and 2 with IC50 = 3.8 and 2.1 nM, respectively, currently in clinical trials. In a whole cell assay, it inhibited PARP activity with EC50 = 4 nM and inhibited proliferation of cancer cells with mutant BRCA-1 and BRCA-2 with CC50 in the 10-100 nM range. MK-4827 was well tolerated in vivo and demonstrated efficacy as a single agent in a xenograft model of BRCA-1 deficient cancer
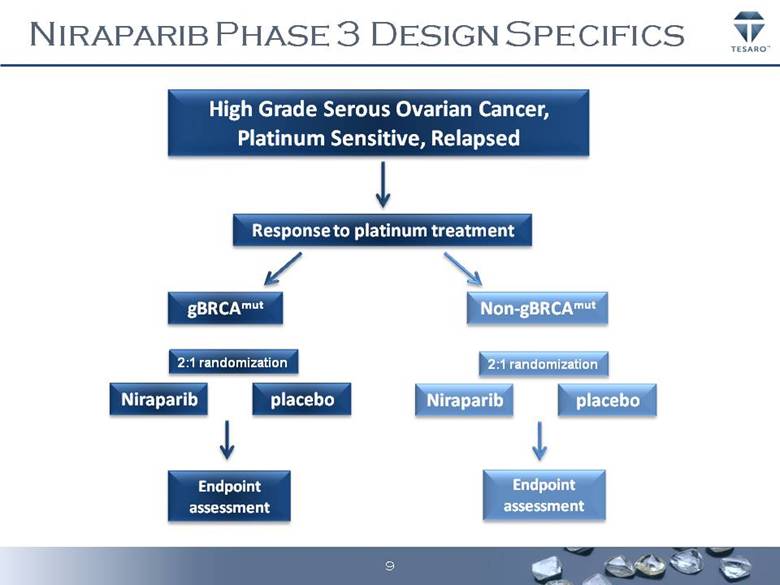
TESARO, Inc. an oncology-focused biopharmaceutical company, today announced that it has initiated patient enrollment in a Phase 3 trial of niraparib, an inhibitor of poly ADP-ribose polymerase (PARP), for the treatment of ovarian cancer. This trial, referred to as NOVA, will evaluate a single daily 300 milligram dose of niraparib in 360 patients with high grade serous, platinum sensitive, relapsed ovarian cancer compared to placebo.
Read more:
| Biological Activity of MK-4827: MK-4827 is a potent, selective, PARP 1/2 inhibitor with IC50 of 3.8 and 2.1 nM for PARP1 and 2, respectively. MK-4827 possesses potential antineoplastic activity. In a whole cell assay, MK-4827 prevented PARP activity with an EC50 of 4 nM, enhancing the accumulation of DNA strand breaks and promoting genomic instability and apoptosis. MK-4827 induces selective synthetic lethality in homologous recombination (HR) repair deficient tumors with BRCA1 / 2 loss and tumor cell lines with non-BRCA-related HR defects, supporting clinical utility in sporadic tumors. MK-4827 reveals good pharmacokinetic properties and is currently in phase I clin. trials. The phase I clinical trials for MK-4827 is ongoing in the treatment of solid tumors. |
| References on MK-4827: 1. Study of the Safety and Efficacy of MK-4827 Given With Temozolomide in Participants With Advanced Cancer (MK-4827-014 AM1).2. A Study of MK4827 in Participants With Advanced Solid Tumors or Hematologic Malignancies (MK-4827-001 AM8). 3. PARP inhibitor MK4827 4. Glendenning J, Tutt A. PARP inhibitors – Current Status and the Walk towards Early Breast cancer. Breast. 2011 Oct; 20 Suppl 3: S12-9. 5. Jones, Philip; Altamura, Sergio; Boueres, Julia et al. Discovery of 2 – {4 – [(3S)-Piperidin-3-yl] phenyl}-2H-INDAZOLE-7-carboxamide (MK-4827): A Novel Oral Poly (ADP-ribose) polymerase (PARP) Inhibitor efficacious in BRCA-1 and -2 Mutant Tumors. Journal of Medicinal Chemistry (2009), 52 (22), 7170-7185. |













Sorry, the comment form is closed at this time.